Global Medical Industry MBA Valuation Lab
The Medical Industry Valuation Lab provides students with interdisciplinary and experiential learning opportunities. Students work in cross-functional teams to conduct market assessments for new medical innovations, evaluate new medical product ideas, and streamline the time to market.
A Valuable Hands-on Experience
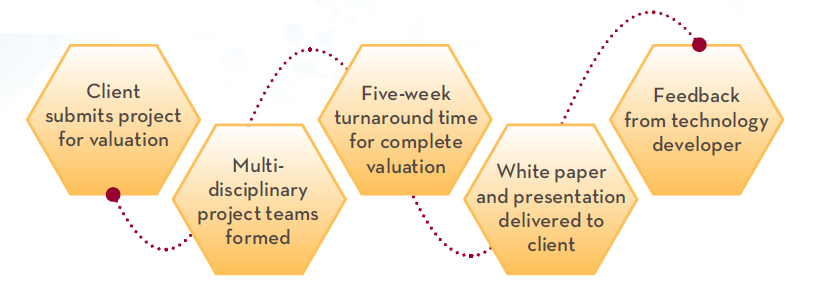
The valuation process involves multiple stages, from assumptions to validations, verifications, challenges, and proposed solutions. This dynamic experience helps students develop a more robust understanding of how to evaluate new technology or service from a global perspective, and help prepare them for leadership roles in an ever-evolving medical industry.
At the end of the 10-day program and intense intellectual inquiry, students prepare client presentations and recommendations for faculty and inventor critique.
The Medical Industry Valuation Lab also provides industry innovators with a top-to-bottom analysis of their medical innovation and its prospects in the market.
In addition, the Medical Industry Valuation Lab is a cross-cultural experience in which students can hone the intercultural skills they have been developing throughout the course of this global program. Students can choose from various global locations, currently including Minneapolis, Palo Alto, Shanghai, and Stockholm. This robust capstone experience is an opportunity for students to explore and network in a foreign city and make lifelong connections with their peers.
In person or online Medical Industry Valuation Lab in Shanghai, Stockholm, Cambridge, or California
Students in this course travel to the Bay Area for ten days of sits visits, tours, lectures, and hands-on project work. During their time in the Bay Area they are exposed to innovations, venture firms, inventors, and medical industry companies. Visits have included: Boston Scientific, IDEO, Kaiser Permanente, Stanford BioDesign, Accenture, Fogarty Institute for Innovation, Schwegman, Lundberg & Woessner Law and others.
Students enrolled into the Medical Industry MBA joint degree program between the University of Minnesota & Tongji University will participate in this course. Students collaborate with their teams to conduct market assessments for new medical innovations. At the end of the 10-day program and intense intellectual inquiry, students prepare client presentations and recommendations for faculty and inventor critique.
Valuation Process
Students will conduct market assessments for new medical innovations in the Medical Industry Valuation Lab, helping evaluate new medical product ideas and streamline the time to market. Working alongside world-renowned faculty, you’ll bring your expertise in market evaluation, industry problems, and cutting-edge policy conversations to the team.
Valuation Components
Through innovative training and industry-specific courses, students will gain knowledge, experiences, and hands-on evaluations of emerging technologies from all around the world.
- Technology
- Market size/potential
- Competition
- Intellectual property
- Regulatory analysis
- Technical evaluation
- User evaluation
- Finance/reimbursement
- Recommendation
Medical Valuation Lab
Hear from Steve Parente, the director of the Medical Industry MBA, and Thom Gunderson, a Valuation Lab council member, during a virtual Q & A session they held about the Medical Valuation Lab with four alumni.

"These lab experiences were pivotal to my career change and ability to lead due to their on-the-ground engagement in different markets and cultures with real-world clients and teams."